Newsdate: June 9, 2025 - 8:30 am
Location: PHILADELPHIA, Pennsylvania
Medicus Pharma Ltd. (NASDAQ: MDCX) ("Medicus" or the "Company") is pleased to announce submission of a comprehensive product development plan to treat external squamous cell carcinoma (SCC) under Investigational New Animal Drug (INAD File No.013880).

Horse isolated in stall.
Company Is advancing use of Novel Doxorubicin-containing Microneedle Array Patch in veterinary oncology as an orphan drug for treatment of external squamous cell carcinoma in horses.
© 2017 by Smerikal New window.
The Company, in December 2024, received a Minor Use in Major Species Designation (MUMS) for its dissolvable Doxorubin-containing microneedle array (D-MNA) to treat external squamous cell carcinoma (SCC) in horses. The product development plan is designed to seek concurrence of the FDA under MUMS designation.
MUMS is a status similar to Orphan Drug status for human drugs. It entitles the Company to an extended 7-year period of exclusive marketing following approval or conditional approval, provided that the Company meets all requirements for maintaining the designation.
Doxorubicin-containing microneedle array (D-MNA) is a patent protected dissovable transdermal patch with cellulose based microneedle arrays that are tip-loaded with doxorubicin. After application, the microneedles function by penetrating the strateum corneum layer of the skin, create a temporary microchannel, penetrate the tumor, dissolve and release doxorubicin into the target tumor, and eradicate the cancer cells.
"In Veterinary Medicine, where there are only a handful of approved oncology drugs, we believe developing a non-invasive treatment for equine SCC represents an untapped and unmet market opportunity potentially in the range of $250 million," stated Dr. Raza Bokhari, Executive Chairman & CEO. "The submission to the FDA of a novel product development program may provide us a first mover advantage and sets the stage to consider other companion animal species and types of cancer."
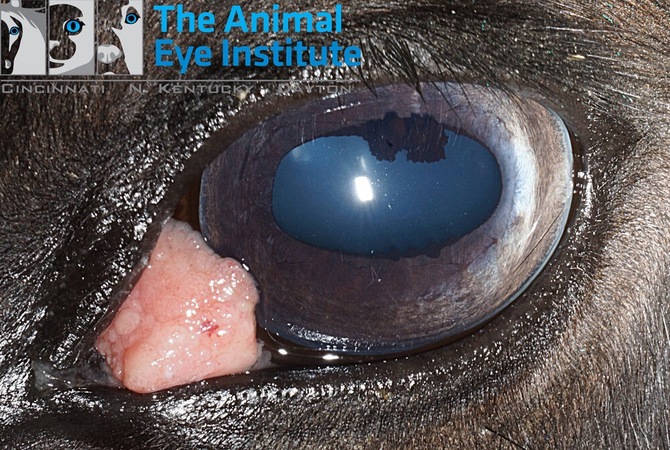
Squamous Cell Carcinoma (SCC) in Horses
SCC is a mucocutaneous skin tumor in horses, primarily affecting adult or aged horses with white or partially white coats. Breeds like Appaloosa, Belgian, American Paint, and Pinto are particularly susceptible. SCC often develops in areas with minimal pigmentation and sparse hair, notably around mucous membranes. More specifically, tumors are mainly seen around the eyes, lips, nose, anus, and external genitalia. The overall incidence of SCC is 2-3% and recent estimates of the US horse population range from 6.6 to 7.25 million horses.p
Current treatment includes surgery to remove the largest mass, cryotherapy of the smaller mass, and local injection of a chemotherapy drug. In some cases, a topical chemotherapeutic drug will be prescribed in the form of drops (tumors near the eye) or cream (tumors in other parts of body). Additionally, some horses are put on an oral drug called piroxicam that may slow the return of the cancerous cells.
The clinical study, SKNJCT-004, is designed to be a randomized, double-blind, placebo-controlled (P-MNA), multi-center study enrolling up to 50 horses presenting with external SCC of the skin. The study will evaluate the efficacy of two dose levels of D-MNA compared to a placebo control.
The horses will be randomized 1:1:1 to one of three groups: a placebo-controlled group receiving P-MNA, a low-dose group receiving 100μg of D-MNA, and a high-dose group receiving 200μg of D-MNA. The horses will be assessed for outcomes over six (6) months from index dose. The primary effectiveness variable will be a veterinarian's assessment for complete clinical response of the target tumor at day 90 according to RECIST.
Click HERE to read complete article.
Press release by Biospace